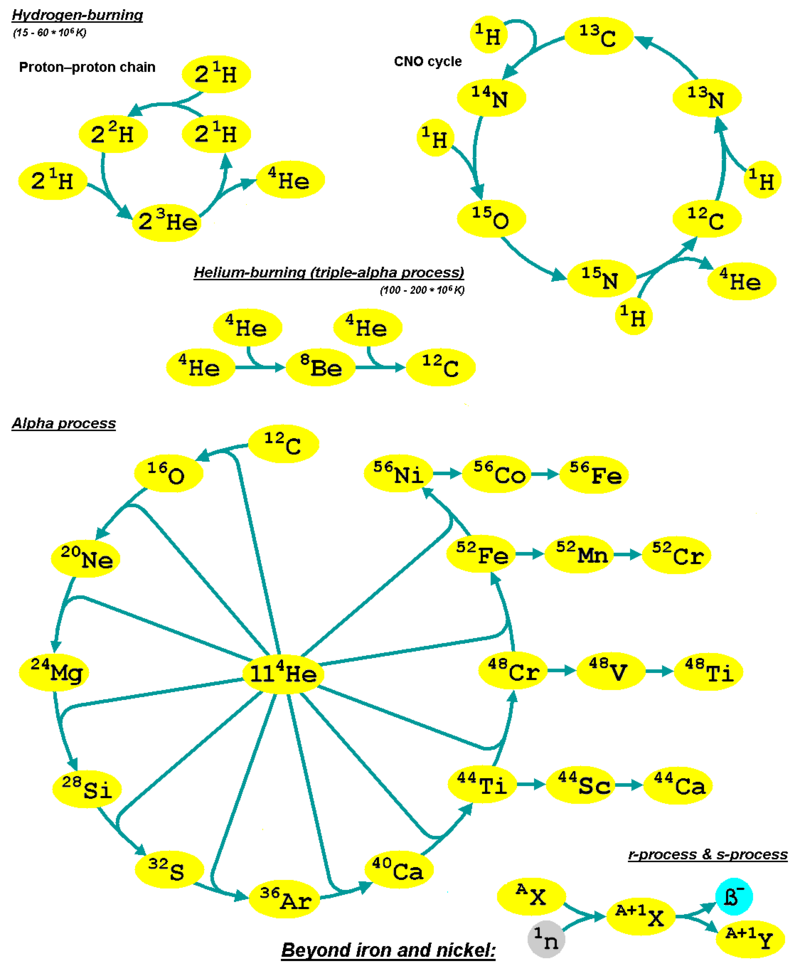
By now, unless you have been living under a rock, you would know that nuclear fusion is the way energy is produced inside the Sun. Most of you would know it is the fusion of hydrogen into helium that produces a huge amount of energy.
But what happens to all the helium produced? Are all stars producing energy through exact same method? How do we get 100s of other elements in the universe if everything started with hydrogen? In this article, we will attempt to answer all of these questions.
Let’s begin!
Hydrogen Fusion
There are primarily three types of hydrogen fusion that occur in the universe.
Deuterium Fusion
This occurs in brown dwarfs, which are sub-stellar objects, also called “failed stars”. They are made from protostars with a mass less than 0.08 times the mass of the sun (Solar mass). Due to low mass (and hence lower gravitational pressure at the core), they are not able to sustain the fusion of hydrogen but can fuse deuterium into helium. Deuterium is a heavy isotope of hydrogen, having 1 neutron and 1 proton in the nucleus, unlike regular hydrogen which has just one proton.
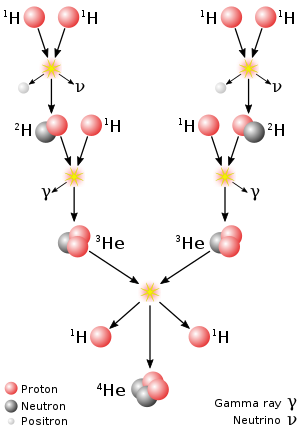
Proton-proto chain reaction
In this, four protons (hydrogen-1) fuse to form a helium-4 nucleus. It is the dominant process that generates energy in the cores of small to mid-size stars like our Sun. In each complete fusion cycle, the proton-proton chain reaction releases about 26.2 MeV.
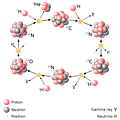
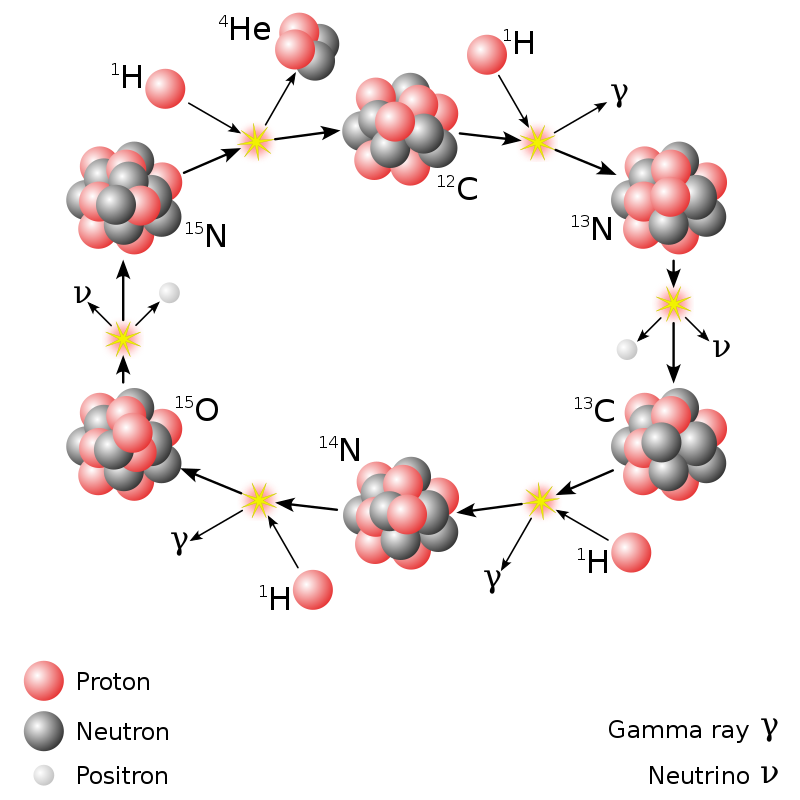
Carbon-Nitrogen-Oxygen Cycle
In higher-mass stars, the dominant energy production process is the CNO cycle, which is a catalytic cycle that uses nuclei of carbon, nitrogen, and oxygen as intermediaries and in the end produces a helium nucleus as with the proton-proton chain. During a complete CNO cycle, 25.0 MeV of energy is released.
Helium Fusion
All the above fusion reactions produced helium at the end. This results in a gradual build of helium at the core of a star. The gravitational pressure at the core, then causes helium to fuse.
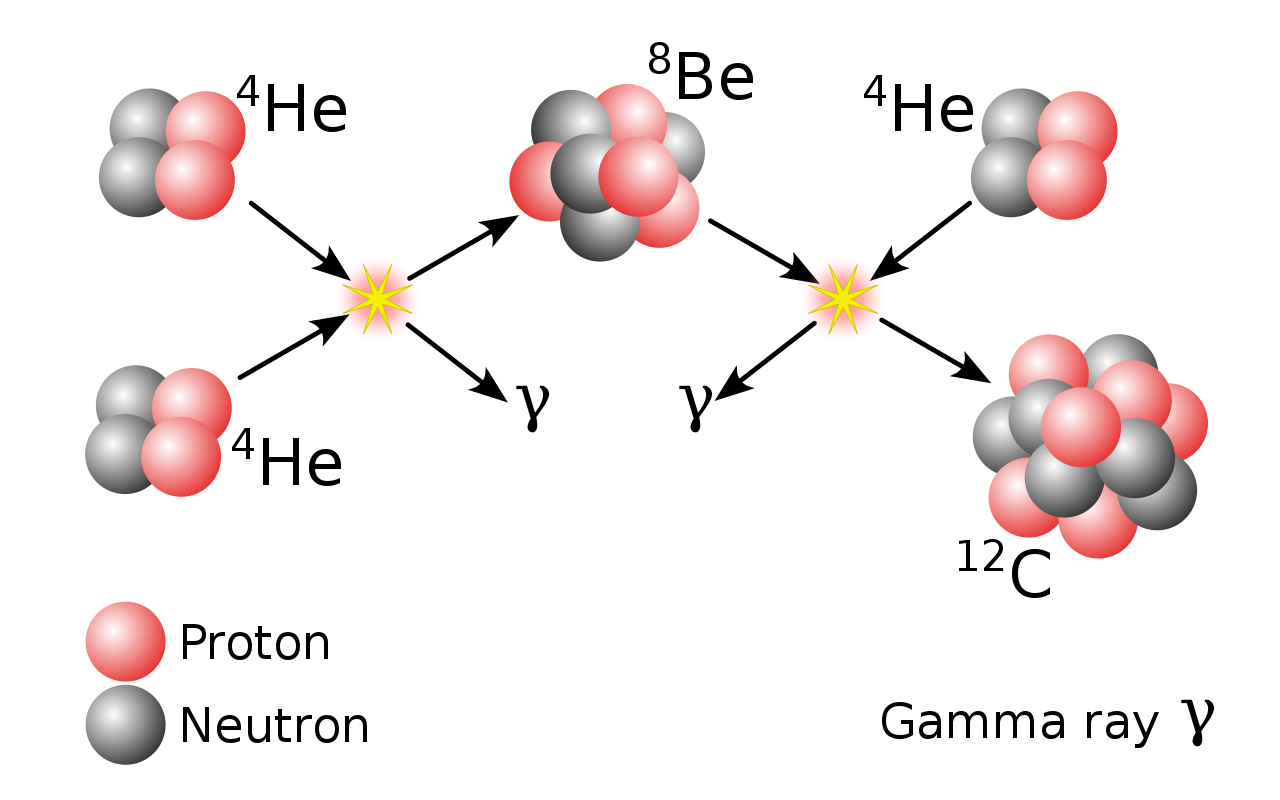
This fusion occurs primarily via two mechanisms: The triple-alpha process and the alpha process.
The triple-alpha process fuses helium into carbon and the alpha process fuses helium into heavier elements up to iron. The alpha process is more common in massive stars.
Neutron-capture process
Sets of nuclear reactions create elements heavier than iron. These reactions occur in a neutron-rich environment of a supernova explosion of a massive star after it reaches the stage of red supergiant. The two common processes are the r and s process.
r-process
The rapid neutron-capture process synthesizes the most neutron-rich stable isotopes of each heavy element. It involves a succession of rapid neutron captures (hence the name) by one or more heavy nuclei, starting with iron.
s-process
The slow neutron-capture process, also known as the s-process, is a set of nuclear reactions that is responsible for the creation of the other half of the heavy elements. The process is slow (hence the name) in the sense that there is sufficient time for this radioactive decay to occur before another neutron is captured.
We believe this article provided a good overview of the complex multitude of nuclear fusion reactions that occur in the universe, in addition to the well-known reaction of fusing hydrogen into helium. It would have also provided you with a basic idea of how so many elements get created in the universe.
In the next article, we will cover another exciting topic related to space and rockets.